On February 25, 2026, we presented our results during on-line seminar organised by Poultry Science Association together with Animal Nutrition Association of Canada. The whole 15 minute presentation can be seen here.
Author Archive: Ivan Rychlik
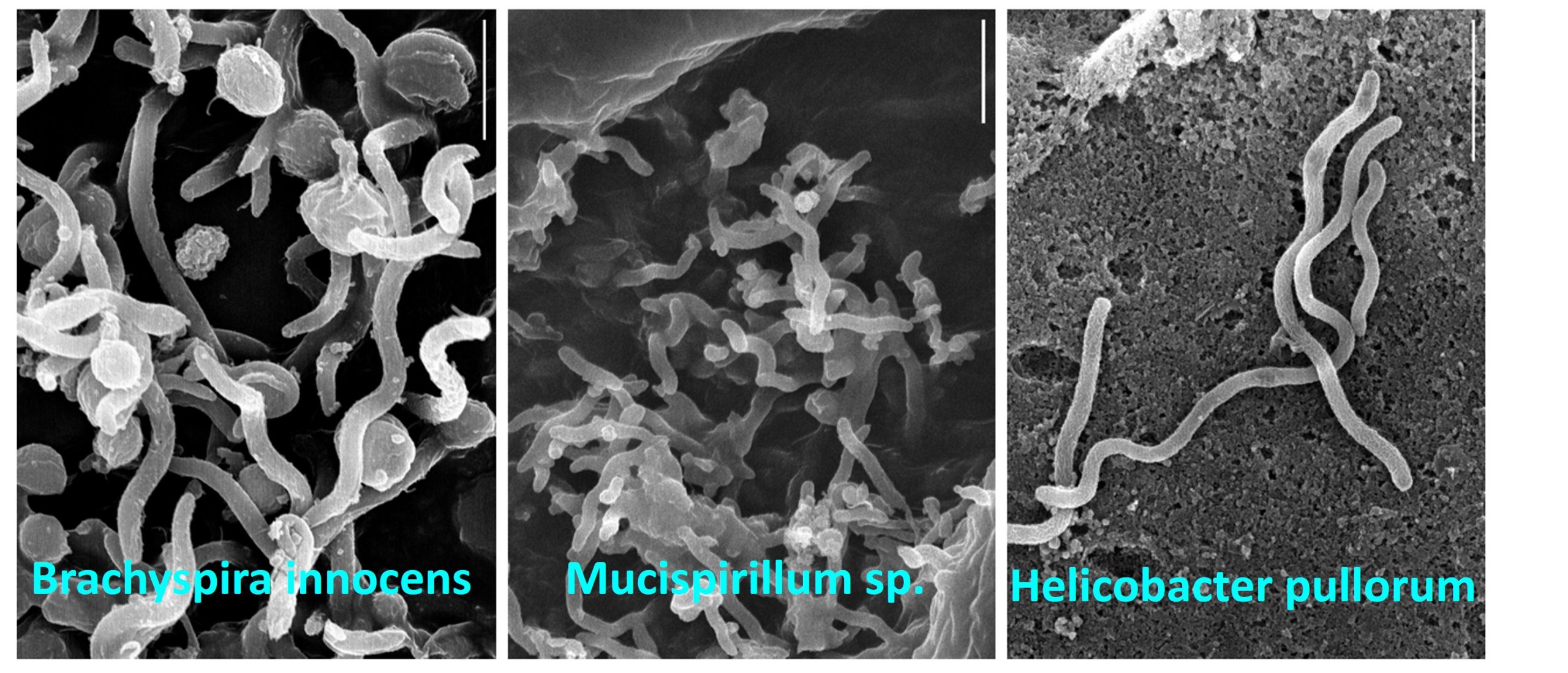
New paper on mucosal microbiota in the chicken caecum
We have just published a new paper in which we define microbiota colonising mucosal surface of chicken caecum. The paper was published in Animal Microbiome and we describe that caecal mucus is colonised by a rather low number of bacterial species from genera Helicobacter, Mucispirillum, Anaerobiospirillum, Desulfovibrio, Brachyspira and Treponema-like spirochaeta. Most of these bacteria are characterisyic by spiral shape of their cells and these bacteria also commonly express type VI secretion system. Additional information including metabolism of bacteria from mucosal surface vcan be found in the paper here.
Probiotics for weaned piglets
We have just published our new findings on novel types of probiotics for weaned piglets. Composition of tested probiotic mixture corresponds with the processes naturally ocurring in gut microbiota of piglets after weaning. This is why the piglets on the day of weaning were provided a defined bacterial mixture in which 5 different species belonging to genus Prevotella dominated. This report should be considered as a pilot one and not as a completed and finalised story. Despite this, it shows how we think and move into an area which has not been explored that much meaning a use of novel types probiotics besides well known lactic acid bacteria. If you view this publication this way, it migth be challenging and it may stimulate you for similar way of thinking and similar experiments. And this is exactly what we wanted.

QuoCNA probiotics for newly hatched chicks in use
On November 26, 2025 additional 180 000 chicks have been treated with QuoCNA probiotics. These chicks will be resistant to Salmonella or E. coli and will enable antibiotic free production. Probiotics were administered using own prototype QuocnaDrop 🙂 More than 6 000 000 chicks have been treated with QuoCNA probiotics in 2025 in the Czech Republic till the end of November.
Publication on new type of probiotics for newborn piglets
A new publication on new type of probiotics for newborn piglets has been published. This study is based on observations that gut microbiota of nursed piglets is dominated by bacterial species from genus Bacteroides. This is why we prepared a mixtures of 4 different species belonging to this genus and used it for oral administration to newborn piglets within first 12 hours after birth. The mixture affected developmenta of gut microbiota and was of overlall positive effect onn health status of treated piglets. Using such intervention we only sped up the process of gut colonisation in newborn piglets and set up all piglets in a litter to the same starting line, i.e. we did not introduce to the production nothing different from natural processes. Currently we develop this approach furthet so that we should be able to introduce a novel type of probiotic veterinary product to common market in 2026.
Influence of monoacylglycerides on chicken resistance to Salmonella
A new paper has been just published. Feed supplementation with a mixture of C1 to C12 monoacylglycerides resulted in lower Salmonella counts at the end of broiler production cycle. Such finding is important for decreased Salmonella transfer from poultry production to human food chain. More information can be found in the original paper published in Poultry Science, either at web page of publisher https://authors.elsevier.com/sd/article/S0032-5791(25)00486-9, or after direct download of pdf file AcylglyceridesPoultrySci.
Active modification of gut microbiota increase chicken resistance to Salmonella
We tested new types of probiotics for chickens in 14 independent experiments. Using hundreds of chickens, efficacy of 22 different mixtures was tested and compared. Unifying feature of the tested mixtures was that all contained different representatives of genus Bacteroides and family Veillonellaceae. Although initial aim of this study was to identify a mixture of the most effective composition, finally we ended up with a conclusiom that if the mixtures contain representatives of genus Bacteroides and family Veillonellaceae, species and strain composition is not that important. The tested mixtures increased chicken resistance to infection with Salmonella approx. 50 times. The more interesting was the effect against spread of Salmonella among chickens by a mere contact. In such experiments, the tested probiotic mixtures decreased spread of infection more than 1000 times. The tested mixtures increased also chicken resistance to Campylobacter jejuni thought the protective effect was not as high as against Salmonella. The paper can be read in full here.
New paper – microbiota vers chicken immunoglobulins
Our new paper is focused of the interactions between immunoglobulin secretion to lumen of the caecum and composition of bacterial microbiota in chickens. To understand the role of chicken immunoglobulins, we used cell sorting of bacteria with and without bound IgA followed by their identification as well as caecal contents originating from wilde type and Ig deficient chickens. Two may conclusion could be drawn. First, bakteria expressing S-layer proteins on their surface bound low levels of chicken IgA. Second, majority of bacteria common to the caecum were highly coated with IgA. IgA secretion was therefore used more important for fixing normal gut flora in chickens rather than to elimination of gut pathogens. For additional information, see the full paper here.
Gut microbiota of autistic and healthy children
Though human gut microbiota does not represent the main research area of our group, we can deal with such topic in a collaboration. A new paper on gut microbiota of autistic and healthy children is just one example. There were significant differences in gut microbiota of these two groups of children although it is difficult to decide whether this differences in gut microbiota were a cause or consequence of autism. We prefer the latter hypothesis, i.e. that changes in gut microbiota of autistic children are consequence and not cause of specific patterns in behavior of autistic children which may include particular food preference. However, even in this case, there is no reason why not to consider interventions that may lead to restauration of “autistic” microbiota to healthy one, being aware of the fact that this will likely be of rather small consequence for total behavioral status of autistic children.
New paper on metabolism of selected anaerobes in the chicken caecum
A new paper on the metabolism of 20 anaerobic bacteria in the caecum of chickens has been published in June 2024. Different species of genus Bacteroides degraded (poly)saccharides. Bacteroides mediterraneensis also expressed type VI secrection system which is used by bacteria for inactivation of competing bacterial species in mixed microbial populations. E. coli or Succinatimonas laso preferred carbohydrate fermentation and the same was true also for Bifidobacterium although carbohydrate fermentation followed a pathways slightly different from reference glycolysis. Megamonas hypermegale and Megamonas funiformis were less dependent on carbohydrate ferementation than previous bacterial species and Megamonas complemented their carbohydrate metabolism by aminoacid degradation. Campylobacter, Sutterella and Phascolarctobacterium degraded aminoacids nearly exclusively with minimal dependence on carbohydrates. Campylobacter expressed flagellar proteins and was therefore motile when colonising chicken caecum. This information can be used for desigh of specific probiotic mixtures competing with particular pathogens not only in the intestinal tract of chickens byt also other farm animals or humans.